In a recent paper in the Journal of Energy Chemistry, researchers discussed the evolution of gas and its relationship to electrode materials in a fast-acting gas monitor for commercial full-size lithium-ion batteries. .
Study: Rapid-operation gas monitor for commercial-sized lithium-ion batteries: evolution of gas and relationship with electrode materials. Image credit: Smile Fight / Shutterstock.com
Contents
Background
Due to their good cycling capacity, high energy density, environmentally friendly, high operating voltage and low self-discharge, lithium ion (LIB) batteries are known to be storage technologies. important energy and have been widely used in electric, portable vehicles. electronics and energy storage systems.
LiNixMnyCo1xyO2 (NMC) based positive electrode materials are suitable for high energy density LIBs; however, they are prone to degradation due to structural and chemical instability. Numerous thorough and exhaustive reviews of gas evolution mechanisms have been published.
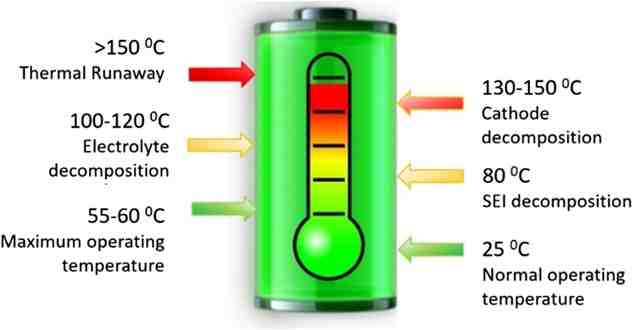
Most gases are studied during the construction of the battery, the first cycle and the phase of thermal leakage, which is based on harsh conditions such as high temperatures above 90 ° C and / or overload. However, the evolution of gases within life-size LIBs in normal operating environments has not been thoroughly described, although it is critical to understand the early stages prior to battery failure or thermal leakage.
About the Study
In this study, the authors discussed the development of a new method for rapid monitoring of gas evolution to address the challenges of implanting multiple gas sensors in commercial batteries, which involved placing non-infrared multigas sensors. dispersive in a sealed tank, where real. commercial batteries with an open end could be put into operation.
The researchers presented a new approach to the continuous monitoring of domestic gases from practical commercial LIBs. Several non-dispersive infrared gas (NDIR) sensors were used in a sealed tank to measure the concentrations of CH4, CO2 and C2H4 during battery operation in a typical design.
Based on the characteristics of non-dispersive infrared (NDIR), this technology allowed the rapid monitoring of operating gas of real commercial batteries, regardless of whether they were bag cells, cylindrical or prismatic. In addition, this methodology was used to capture the quantitative consumption of CO2 throughout the discharge process in real batteries, as well as the interaction between CO2 and Li2CO3.
The team developed a model to separate the effects of voltages and temperatures on the development of oxygen. The role of stress in the evolution of CO2 was assessed. In addition, the evolutionary behaviors of CH4, CO2, and C2H4 were quantified and the impact of operating temperature variation on the link between CH4 and C2H4 was illustrated. The concentration and rate of evolution of the CO2 in the electrolyte were also modeled near the edge of the electrolyte (point V).
Observations
The concentration of CO2 produced was closely related to stress and temperature, but the concentrations of CH4 and C2H4 depended solely on temperature. The CO2 concentration was higher and the rate of evolution was faster when the temperature rose to 40 ° C and the higher cut-off voltage was raised to 4.5 V. During the second discharge process, it was consume C2H4 and the concentration dropped. less than 6500 ppm. The C2H4 concentration remained stable at 18000 ppm until the solid electrolyte interface (SEI) was stable, but CH4 increased to 60400 ppm. When the cut-off voltage was increased to 4.5 V, O2 showed a similar diffusion behavior. Below 4.5 and 4.6 V, the rate of evolution of CO2 changed substantially and the value approached 2 mol m-3 s-1.
More from AZoM: Innovating for the Earth – World Earth Day 2022 at AZoM
At 0.1 s, the rate of O2 creation was about -0.359 mol m-3, while the rate of CO2 generation was about -182.713 mol m-3. CO2 concentration and peak generation peaks occurred at 1.25 and 2.75 seconds, respectively. The existence of Li2CO3 was indicated by a peak near 55.2 eV in the 1s spectra. In the C 1s spectra, a prominent peak was recorded at about 290 eV as Li2CO3. The peaks between 531, 8 and 532, 9 eV in the O 1s spectra were assigned to C = O of Li2CO3 and C – O, respectively.
CO2 was associated with the creation of O2 from a positive electrode LiNi0.5Mn0.3Co0.2O2 as a typical trace gas, which meant a stable emission of CO2 below a threshold voltage of 4.5 V During the discharge process at different cutting temperatures and stresses, a larger amount of Li2CO3 was observed on the surface of the graphite negative electrode.
Conclusions
In conclusion, this study elucidated a unique approach based on the use of multiple NDIR gas sensors that can be used to monitor the internal gas concentration of commercial LIBs in real time without affecting battery performance. Temperature was found to have no effect on O2 diffusion or CO2 concentration. High voltage, on the other hand, accelerated the rate of electrochemical reaction at the electrode-electrolyte interface, leading to an increase in CO2 concentration.
The authors mentioned that this method could be used to see the internal evolution of gas in batteries and help to understand the mechanism of gas revolution. They believe that the proposed operating techniques promise to provide a basis for a better understanding of the interaction of gas-related side reactions between positive and negative electrodes in commercial LIBs.
Source
Zhang, J., Zhang, X., Bi, S., Rapid-operation gas monitor for commercial life-size lithium-ion batteries: gas evolution and relationship with electrode materials. Journal of Energy Chemistry (2022). https://www.sciencedirect.com/science/article/abs/pii/S2095495622002005
& # xD;
Disclaimer: The views expressed herein are those of the author expressed in private and do not necessarily represent the views of AZoM.com Limited T / A AZoNetwork the owner and operator of this website. This disclaimer is part of the Terms and Conditions of Use of this website. & # XD;
How do you take care of LiPo batteries?
Q: Are lithium polymer batteries safe? A: Yes. Lithium polymer is even safer than lithium ion, as there is less risk of filtering the electrolyte component.
- How to Store LiPo Batteries
- Never store loose batteries together. …
- Never store batteries in extreme temperatures or in direct sunlight. …
- Always disconnect batteries when not in use, and store them in a non-conductive, flame-retardant container.
Never damage, puncture, or impact batteries or related components.
Is it OK to leave LiPo batteries fully charged?
What not to do with a LiPo battery? Never charge a packed LiPo that has been inflated or inflated due to excessive or lower load or shock. Never charge a lithium polymer battery that has been punctured or damaged in a crash. Never charge a LiPo battery while it is inside your model or other electronic device.
What happens if you leave a LiPo battery fully charged?
Do LiPo batteries need to be discharged?
We kept them dead. But you shouldn’t do this with LiPo batteries. LiPo batteries should not be stored at full charge either. For longer battery life, LiPos should be stored at room temperature at 3.8 V per cell.
How long can I keep a fully charged LiPo?
LiPo batteries also do not develop memory or voltage drop features such as NiCd / NiHm batteries, and do not need to be discharged before charging. But they are not exempt from their disadvantage. Improper handling of these batteries can result in fire, explosion, and inhalation of toxic fumes.
Do lithium-ion batteries have sulfuric acid?
After 3 months, the battery capacity is reduced to 80%, so the package is defective (100% damage). 3 months is about 100 days, so every day of storage at full charge voltage does 1% damage.
The positive and negative electrodes of lead-acid batteries are lead oxide, lead metal, and the electrolyte is concentrated sulfuric acid. The positive and negative electrodes of the lithium battery are cobalt lithium / lithium iron phosphate / lithium manganate, graphite, organic electrolyte.
What is the main ingredient in lithium-ion batteries?
Do batteries produce sulfuric acid? The electrolyte solution of lead acid batteries contains sulfuric acid, which is highly corrosive and can cause severe chemical burns to the skin and damage the eyes. The solution is also poisonous if ingested. In addition, overcharging a lead acid battery can produce hydrogen sulfide gas.
What is meant by lithium-ion battery?
The most common combination is lithium (cathode) and graphite (anode) cobalt oxide, which is most commonly found in portable electronic devices such as cell phones and laptops. Other cathode materials include lithium manganese oxide (used in electric and hybrid electric cars) and lithium iron phosphate.
What is lithium-ion battery and how does it work?
A lithium-ion (Li-ion) battery is an advanced battery technology that uses lithium ions as a key component of its electrochemistry. During a discharge cycle, the lithium atoms of the anode ionize and separate from their electrons.
What are lithium-ion batteries used in?
A lithium ion battery is a type of rechargeable battery that is charged and discharged by lithium ions moving between the negative (anode) and positive (cathode) electrodes.
What batteries contain sulfuric acid?
Lithium-ion batteries provide portable electricity, powering electronic devices such as cell phones, laptops, and tablets. Lithium-ion batteries are also used to supply power to medical equipment, electric vehicles, and power tools.
Is sulfuric acid in AA batteries?
A lead-acid battery consists of two lead plates separated by a liquid or gel containing sulfuric acid in water. The battery is rechargeable, with chemical charging and discharging reactions.
Do all batteries have sulfuric acid?
Car batteries are usually lead-acid batteries and contain sulfuric acid. The sulfur in a lead-acid battery is very corrosive. Diluted sulfur is sometimes used topically to treat acne and other skin conditions, but battery acid sulfur is not diluted enough to be safe for the skin.
Do lithium-ion batteries contain acid?
When a lead acid battery is fully charged, the electrolyte is made up of a solution consisting of up to 40 percent sulfuric acid, and the rest consists of normal water. As the battery discharges, the positive and negative plates gradually become lead sulfate.
Which batteries contain acid?
Lithium ion is a low maintenance system, an advantage that most other chemicals cannot claim. There is no memory and the battery does not require scheduled cycles to extend its life. Lithium ions also do not have the problem of sulfation of lead acid that occurs when the battery is stored without periodic charging.
What is inside a lithium-ion battery?
Battery acid is sulfuric acid that has been diluted with water to achieve a concentration level of 37%. This particular type of acid is used in sealed lead acid batteries, however, concentration levels differ with some brands.
What is the difference between lithium-ion battery and lead acid battery?
The basics. A battery consists of an anode, cathode, separator, electrolyte and two current collectors (positive and negative). The anode and cathode store lithium. The electrolyte carries positively charged lithium ions from the anode to the cathode and vice versa through the separator.
When should I charge my lithium-polymer battery?
Charging a lead-acid battery can take more than 10 hours, while lithium-ion batteries can take up to 3 hours to a few minutes to charge, depending on the size of the battery. Lithium ion chemicals can accept a faster current rate, charging faster than lead-acid batteries.
Fully charge your package before first use. Charge the LiPo battery to 5C or less with LiPo settings only. You should use a balancing charging system similar to the Hyperion, Hitec, EV-Peak or Graupner chargers offered here at MaxAmps.
When should I charge my LiPo battery?
Is it okay to leave a lithium polymer battery in the charger? Lithium ion batteries can be used up to 20% of their capacity. Unlike lead acid batteries, it will not damage the battery to use the chance charge, which means that a user could plug in the battery during a lunch break to complete the charge and finish their shift without the battery running out. go down too much.
Should I discharge LiPo battery before charging?
LiPo batteries are fully charged when they reach 4.2 v / cell, and their minimum safe charge, as we will discuss in detail later, is 3.0 v / cell. 3.7 v is practically in the middle, and this is the nominal load of the cell.
How often should you balance charge a LiPo?
LiPo batteries also do not develop memory or voltage drop features such as NiCd / NiHm batteries, and do not need to be discharged before charging. But they are not exempt from their disadvantage. Improper handling of these batteries can result in fire, explosion, and inhalation of toxic fumes.
Can you charge a LiPo battery right after use?
But my 1/8 buggy balances them every time it loads, because one cell in all my packages always seems to pull one cell lower than the rest. A good battery should be able to maintain a good balance between the cells for a month without any problems. But if your charger already has a built-in balancer, you can also use it.
Should LiPo batteries be discharged before charging?
It is recommended to let it cool first. No problem recharging immediately. Packages will continue to cool during the upload process.
What happens if you fully discharge a LiPo battery?
LiPo batteries also do not develop memory or voltage drop features such as NiCd / NiHm batteries, and do not need to be discharged before charging. But they are not exempt from their disadvantage. Improper handling of these batteries can result in fire, explosion, and inhalation of toxic fumes.
How often should you discharge LiPo batteries?
Excessive discharge (continuing to use a LiPO battery even when it is almost completely depleted or below its minimum voltage level) can cause permanent damage to the battery. Damage caused by this may be INTERNAL damage to the battery and will not always be visible.
Why do you discharge a LiPo battery?
Never leave LiPo batteries fully charged for more than 2 or 3 days. If you notice that you will not be using the battery today on the third day, you will need to discharge it to 3.6 v-3.8 v per cell for safe storage until you are ready to use the battery again.
Do batteries give off gas?
They became weak and have a good ability at low currents, they are powerless to give energy; the voltage drops after 30-60 seconds. even if they are fully charged. This is the only reason to unload them and store them in a cool place (above freezing).
When the batteries are recharged, they generate hydrogen gas which is explosive at certain concentrations in the air (the explosive limits are 4.1 to 72 percent hydrogen in the air). The ventilation system can exchange an adequate amount of fresh air for the number of batteries being charged.
Are battery fumes harmful?
What gas does a battery emit? Gases that are released when the batteries are being charged – hydrogen (highly flammable and easily ignited) and oxygen (supports combustion) – can cause an explosion. The acid used as an electrolyte in batteries is also very corrosive and can cause injury if it comes in contact with workers.
What voltage is MX fuel?
Batteries, found in billions of consumer devices such as smartphones and tablets, were found to leak more than 100 toxic gases, including carbon monoxide. Gases, which are life-threatening, can cause severe irritation to the skin, eyes and nasal passages, and damage the environment.
What is MX fuel?
Milwaukee MX FUEL Batteries MX FUEL CP203 Battery: This “compact” MX FUEL battery pack includes twenty (20) 3.6 V 21700 lithium ion cells arranged (roughly) in two layers. Effectively get a 3 Ah package to operate MX FUEL tools and equipment.
What battery fits Milwaukee?
| MX FUEL, an innovative wireless lightweight equipment system, goes beyond the limitations of gasoline and power cord units. The MX FUEL equipment system redefines the light equipment market by offering the performance, execution time and durability required by the trades. | Number of batteries |
|---|---|
| You need 2 C batteries (included) | Voltage |
Do batteries give off carbon monoxide?
18 Volts
Is battery low or carbon monoxide?
Lead batteries were determined not to emit carbon monoxide. However, they emit hydrogen. It was also noted that carbon monoxide detectors will be activated when exposed to large amounts of hydrogen. Hydrogen gas such as carbon monoxide is a colorless, odorless gas.
What can cause false carbon monoxide readings?
Here’s a simple guide: Smoke alarms alert you with three beeps in a row. Carbon monoxide alarms alert you with four beeps. A single whistle means the battery is low or the detector needs to be replaced.
What gas does a lead acid battery give off?
CO alarms become erratic once they expire. This is the most common cause of false alarms. Excessive humidity in a bathroom can trigger the CO alarm. CO alarms should not be installed in areas with excess steam.


Comments are closed.